Clinical Trial Content Examples Generated from Protocols
Clinical Trial Content Examples
Generated from Protocols
Explore real clinical trial documents generated from study protocols including patient-facing materials, operational summaries and feasibility reports.

Clinials generates a wide range of clinical trial documents directly from study protocols. From operational summaries and lab billing reports to patient recruitment and investor-ready outputs, each document is structured, traceable, and review-ready.
Clinials generates a wide range of clinical trial documents directly from study protocols. From operational summaries and lab billing reports to patient recruitment and investor-ready outputs, each document is structured, traceable, and review-ready.
Detailed Patient Informed Consent Form (P.I.C.F)

Transform complex clinical trial protocols into clear caregiver education materials that explain study goals, procedures, risks, and the participant journey in simple language.
Brief Participant Information and Consent Form (P.I.C.F)

Transform complex clinical trial protocols into clear caregiver education materials that explain study goals, procedures, risks, and the participant journey in simple language.
Regulatory & Quality Documents
Regulatory & Quality Documents
AI-generated documents to support regulatory compliance, quality oversight, and trial readiness for Sponsors, CROs, and Sites.
AI-generated documents to support regulatory compliance, quality oversight, and trial readiness for Sponsors, CROs, and Sites.
Operational Protocol Documentation for Clinical Trials
Operational Protocol Documentation for Clinical Trials
AI generated operational reports used by CROs, Sponsors and Sites
AI generated operational reports used by CROs, Sponsors and Sites
Operational Protocol Synopsis

A structured operational protocol synopsis summarising study design, objectives, endpoints, procedures, and operational requirements for study teams.
Patient Information Sheet

Plain-language patient information sheets generated directly from the clinical trial protocol, covering study purpose, risks, visits, and participation requirements.
Protocol to Marketing Report

Protocol to Marketing transforms clinical trial protocols into compliant, patient-friendly marketing materials that support recruitment, feasibility, and stakeholder communication.
Patient Information Sheet

Plain-language patient information sheets generated directly from the clinical trial protocol, covering study purpose, risks, visits, and participation requirements.
Participant Educational Brochure

Explains study procedures. risks, participant protections, and emphasizes voluntary participation
Caregiver Educational Brochure

Transform complex clinical trial protocols into clear caregiver education materials that explain study goals, procedures, risks, and the participant journey in simple language.
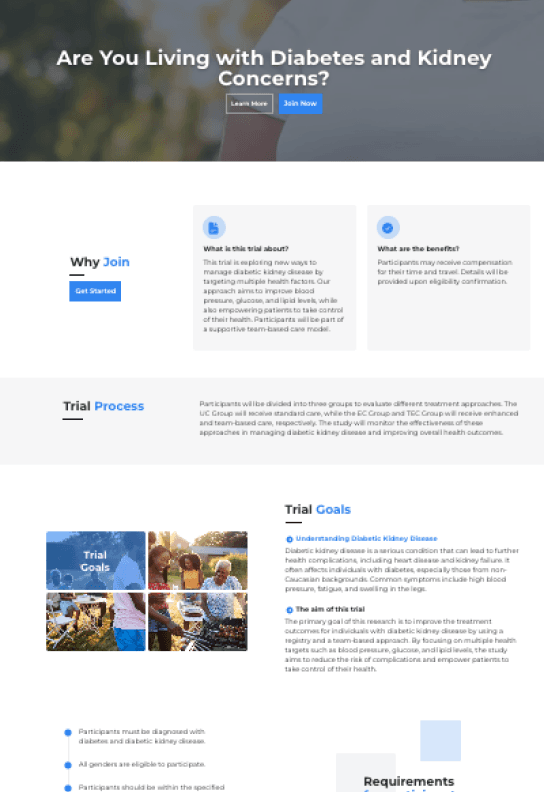
Patient Recruitment Materials Generated from Protocols
Patient Recruitment Materials Generated from Protocols
Create high-performing landing pages & recruitment materials in seconds.
Create high-performing landing pages & recruitment materials in seconds.
Clinical Trial Feasibility & Planning Reports
Clinical Trial Feasibility & Planning Reports
Clinical Trial Feasibility & Planning Reports
Create reports that assess whether a study can be run successfully at a site.
Create reports that assess whether a study can be run successfully at a site.
Patient Visit Timeline

Timeline that includes the key visits and all the timepoints that are required for the patient
Budget Discovery

A protocol-derived breakdown of study activities, assessments, biomarkers, and endpoints to support feasibility and early budget planning.
Schedule of Activities

A complete Schedule of Activities from clinical trial protocols, including biomarkers, procedures, and endpoints organised by visit to help sites and sponsors plan study execution and budgets.
Laboratory Report

A protocol-derived, fully traceable list of lab tests — structured for review, feasibility, and lab planning.
Laboratory Reports for Clinical Trial Billing
Laboratory Reports for Clinical Trial Billing
Create reports that identify all the laboratory tests in a protocol.
Create reports that identify all the laboratory tests in a protocol.
Investor & Budget Discovery Reports
Investor & Budget Discovery Reports
Clinials transforms complex clinical trial documents into easy-to-read, investor-ready output.
Clinials transforms complex clinical trial documents into easy-to-read, investor-ready output.
Investors Trial Overview

Protocol-derived trial information for investment evaluation.