For too long, clinical trial teams have manually translated complex protocols into operational plans. The Clinials Schedule of Activities report changes that calculus entirely, bringing precision, traceability, and real-time clarity to one of the most operationally demanding tasks in trial management.
Clinical trials are, at their core, exercises in disciplined complexity. Every protocol defines a precise choreography of assessments, procedures, and administrative actions, each tied to a specific cohort, a specific timepoint, and a specific intent. Yet the tools most site teams have relied upon to operationalise this choreography have lagged far behind the sophistication the work demands.
Spreadsheets built manually. Tables copied painstakingly from PDF protocols. Version conflicts resolved in email threads. The gap between what a protocol specifies and what a site team can readily act upon has, for years, represented one of the industry's most quietly costly inefficiencies.
At Clinials, we built the Schedule of Activities (SOA) report to close that gap. And in doing so, we have created something that goes well beyond a better-formatted table, we have created a live, protocol-grounded operational document that scales with your trial.
What the Schedule of Activities Report Delivers
The Clinials SOA report is a structured, auto-generated output that extracts the full schedule of trial activities directly from your protocol, organising them intelligently by cohort and timepoint. It is not a static export. It is a dynamic reflection of your protocol's intent, rendered in an operationally actionable form.

Here is what the report outputs, and why each element matters:
Core Report Features
Assessments grouped by cohort and timepoint, structured to mirror how site teams actually plan and execute work, reducing the cognitive overhead of cross-referencing protocol sections
Ordered by cohort and timepoint, ensuring logical sequencing that aligns with visit workflows and eliminates manual sorting
Activity type classification (Assessment, Admin, Procedure, etc.) enabling teams to distinguish clinical assessment work from administrative tasks, facilitating more precise workload planning
Quantity fields based on trial target numbers allowing budget holders and coordinators to understand resource demand at a per-activity, per-cohort level
Protocol citations embedded every activity traces back to its source in the protocol, supporting audit readiness, regulatory inspection, and deviation management
Export to Excel for seamless integration into existing operational workflows, cost models, and site management systems
Multi-language translation when a different language is requested, the entire report translates, enabling global multi-site trials to operate in local languages without duplicated effort
A Glimpse of the Output
To make this concrete, below is a representative illustration of what the SOA report produces — structured, typed, cited, and quantity-aware:
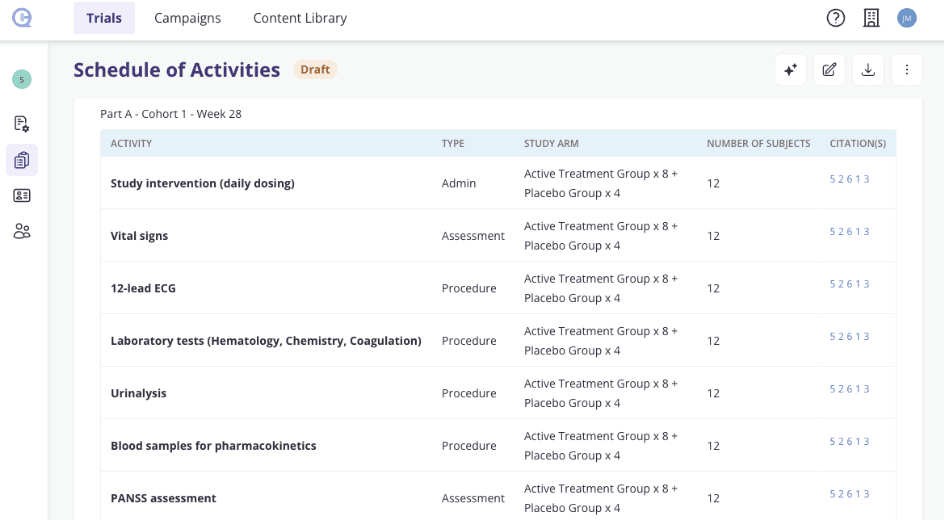
Every row is generated from the protocol. Every citation is traceable. Every quantity reflects the trial's target enrolment numbers. This is not a manual table it is a living document that changes as the protocol changes.
"The distance between what a protocol says and what a site team can act on has always been a source of risk. The Schedule of Activities report eliminates that distance."
Why This Matters for Clinical Operations
Protocol Fidelity at the Point of Execution
One of the most persistent risks in clinical trial execution is protocol deviation often not from negligence, but from the simple difficulty of keeping an operationally-translated plan aligned with the source document. When site coordinators are working from a manually-built spreadsheet that was transcribed two protocol amendments ago, misalignment is not a failure of diligence; it is a failure of tooling.
The Clinials SOA report addresses this structurally. Because every activity in the output carries a direct citation to its protocol section, any divergence between the report and the protocol is immediately apparent. This supports both proactive quality management and reactive audit response with teams able to demonstrate, at any moment, the exact basis for every scheduled activity.
Operational Planning at Scale
The inclusion of quantity fields calculated against trial target enrolment numbers transforms the SOA from a compliance document into a planning instrument. A study coordinator can look at Cohort B, Week 8, and know immediately that 62 ECGs, 124 blood draws, and 62 PRO questionnaires are required. A budget holder can interrogate that data to build accurate site cost models. A clinical operations lead can use it to forecast staffing requirements across multiple sites.
This level of operational granularity, derived automatically from the protocol, has historically required hours of manual calculation. The SOA report delivers it in seconds.
Activity Type Classification: The Underappreciated Feature
The inclusion of activity type, Assessment, Admin, and Procedure is a feature that may appear straightforward but has profound downstream impact. Clinical research teams are rarely homogeneous in their responsibilities. Assessors, coordinators, and proceduralists often have distinct workflows, distinct training requirements, and distinct scheduling constraints.
When a site team can filter the SOA by activity type, they can immediately extract the workload relevant to each role. The assessment team sees their tasks. The administrative staff see theirs. There is no ambiguity, no manual segmentation, no risk of a critical procedure being attributed to the wrong workflow.
4. Multi-Language Support: Global Trials, Local Clarity
For sponsors running multi-regional trials, the translation capability of the SOA report is not a convenience it is an operational necessity. Regulatory submissions, site training documents, and operational guides must frequently be produced in the local language of each participating country. The capacity to generate a protocol-cited, fully translated SOA in a single workflow step removes a significant bottleneck from global trial operations and reduces the risk of translation-introduced error.
5. Excel Export: Meeting Teams Where They Work
The ability to export to Excel reflects a pragmatic understanding of how clinical trial teams operate. We do not ask teams to abandon their existing systems; we provide outputs that integrate cleanly with the tools they already use for cost modelling, for site communication, for regulatory filing preparation. An Excel-formatted SOA that carries protocol citations can be embedded directly into a site initiation package, a monitoring plan, or a budget justification with no reformatting required.
The Broader Strategic Imperative
The clinical trials industry is under increasing pressure to do more with less. To accelerate timelines, reduce per-trial costs, and maintain the quality standards that protect participant safety and data integrity. Technology that addresses only one of these imperatives at a time is insufficient. The tools that will define the next era of clinical operations will be those that simultaneously improve speed, reduce risk, and enhance clarity.
The Clinials Schedule of Activities report is built to that standard. It is faster than manual transcription by orders of magnitude. It is more reliable because it eliminates the human error inherent in manual extraction. And it is clearer because it organises complex protocol requirements into a structure that site teams can immediately interpret and act upon.
In a field where the cost of ambiguity is measured in protocol deviations, regulatory findings, and most critically participant outcomes, this kind of operational clarity is not a luxury. It is a baseline requirement.
An Invitation to Rethink the Standard
If your team is still building trial schedules manually extracting activities from PDF protocols, maintaining parallel spreadsheets, and reconciling versions across amendment cycles, I would encourage you to consider what that overhead is truly costing you. Not just in hours, but in quality. In risk. In the attention that could be directed toward the participants your trial is designed to serve.
The Clinials Schedule of Activities report is available now. We would welcome the opportunity to demonstrate how it fits within your trial management workflow.
The protocol already contains the plan. Clinials makes it operational.