Clinials helps medical device companies accelerate clinical readiness by transforming complex protocols into structured, compliant, and operationally usable intelligence reducing friction across regulatory, clinical, and site-facing workflows.
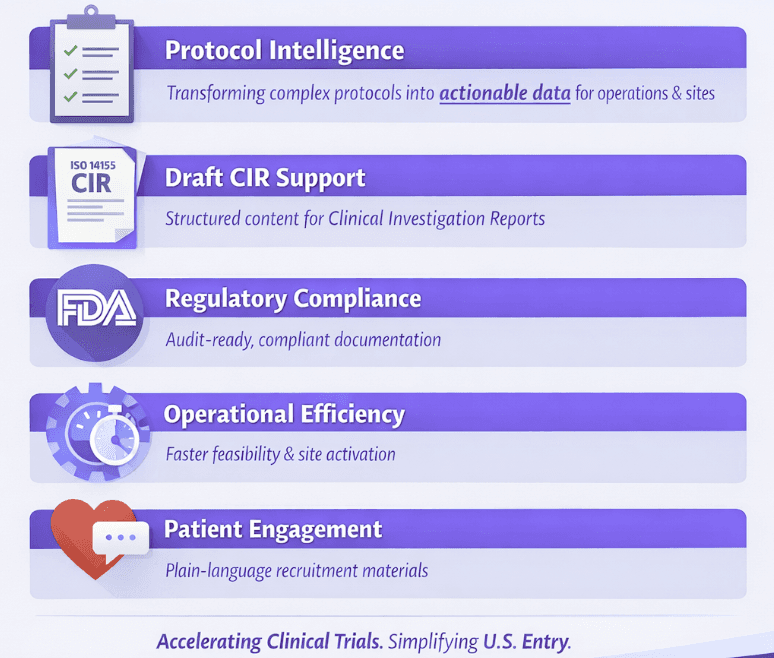
Protocol Intelligence: To Actionable Outputs
Device protocols are dense, technical, and highly scrutinised. Clinials extracts structured data, objectives, endpoints, eligibility criteria, safety frameworks, device descriptions, and statistical plans, and converts it into:
Operational summaries for sites
Feasibility and budget planning inputs
Patient-facing materials in plain language
Cross-functional briefing documents
This improves clarity and alignment across U.S. clinical teams, CROs, and investigators.
Draft Clinical Investigation Report (CIR) Support
Aligned with ISO 14155
Clinials extracts protocol content to form the structured backbone of a draft Clinical Investigation Report.
This enables:
Early report framework development
Consistency between protocol and final report
Improved traceability and audit readiness
Reduced medical writing rework
For companies preparing IDE, PMA, or 510(k)-supporting clinical documentation, this consistency reduces regulatory friction.
Compliance-Built AI (Not Generic AI)
Clinials is designed specifically for regulated clinical research environments. Outputs maintain alignment with source documents, helping sponsors meet U.S. regulatory expectations where documentation precision and consistency are heavily scrutinised.
Operational Efficiency for U.S. Trials
Entering the U.S. market often exposes inefficiencies in document interpretation, site onboarding, and cross-team communication.
Clinials reduces:
Manual protocol re-interpretation
Reformatting across departments
Misalignment between regulatory and operational documentation
This accelerates feasibility, site activation, and trial start-up.
Patient Communication and Recruitment
U.S. regulators increasingly expect clarity and accessibility in participant materials. Clinials generates plain-language summaries and recruitment-ready content directly from the protocol, improving comprehension and supporting more representative enrolment.
The Strategic Advantage
For medical device companies entering the U.S. market, the challenge is not just regulatory approval, it’s operational precision and documentation consistency under scrutiny.
Clinials turns the protocol into structured intelligence that supports:
Faster study planning
Cleaner regulatory documentation
Stronger site engagement
Earlier CIR readiness
Reduced compliance risk
In short: fewer manual bottlenecks, more regulatory confidence, and accelerated path to U.S. market entry.